Why kidney disease damages the vascular system
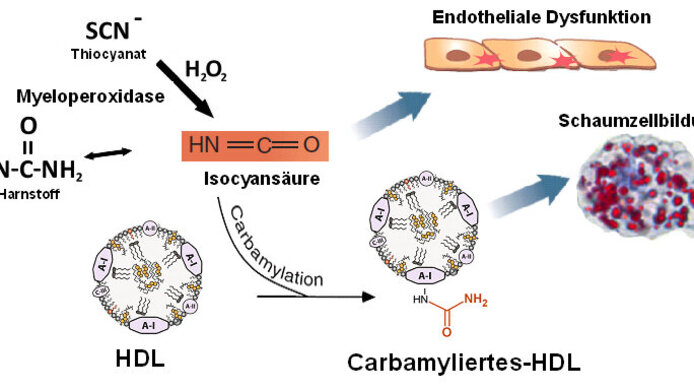
Patients suffering from chronic renal disease frequently die from cardiovascular complications. A damaged and poorly functioning vascular system is the reason for such complications. A team working with Gunther Marsche at the Medical University of Graz on a project supported by the FWF investigated the extent to which a specific decomposition product (cyanate) of urea, which occurs in a concentrated form in the blood of patients with renal disease, is responsible.
One team – two discoveries
“We were able to identify two previously unknown mechanisms in human tissue and in an animal model by which cyanate can lead to a dysfunctional vascular system in patients with chronic renal disease”, explains Gunther Marsche. “In the first mechanism, cyanate causes an adverse change in a lipoprotein, which, in its unchanged state, is key to the healthy functioning of blood vessels. In the second case, cyanate activates a gene that can amplify inflammatory responses in the vascular system.”
From good to bad cholesterol
The lipoprotein that is modified by cyanate is the High Density Lipoprotein (HDL), also known as “good” cholesterol. As the team working with Marsche was able to demonstrate, HDL undergoes a chemical change known as carbamylation. The results are drastic, as Marsche outlines: “While, prior to this modification, HDL helped to prevent vascular constriction, this protective effect is dramatically reduced by carbamylation. In extreme cases, it can even be a factor in vascular constriction.” This is an effect that can occur particularly in patients with chronic renal disease. This is because they excrete urea considerably less effectively than healthy individuals and, as a result, the decomposition product, cyanate, is present in significantly higher concentrations in the blood.
Gene activation
The second effect that cyanate has on the vascular system and which was identified by Marsche’s group, is caused by the activation of a gene. This codes for a so-called adhesion molecule, which is located on the cell membrane of the endothelial cells, i.e. on the cell layer that lines a vessel. The protein known as ICAM-1 functions by pointing certain blood cells (leucocytes) “in the right direction” to fight infections or tumours. In addition, leucocytes have receptors that bind ICAM-1. Marsche was able to now show that cyanate’s activation of the gene for ICAM-1 leads to increased binding of leucocytes, and inflammatory responses of the vessel walls possibly be exacerbated. This chain of effects establishes for the first time a causal link between uraemia (an excess of urea in the blood), often seen in patients with renal disease, and inflammatory responses.
From knowledge to treatment
The findings from this FWF project are urgently needed, as the average age of the general population increases and with it the number of patients with chronic renal disease. Their mortality increases sharply in particular as a result of secondary cardiovascular disease. To date, however, little is known about how these sequelae occur. For Marsche, the fundamental importance of these discoveries within the framework of the FWF project is therefore clear: “The data from this FWF project provides an approach for the clinical evaluation of substances that counteract cyanate in the blood. This could lead to the development of medications and treatments that would facilitate a much better quality of life for patients with renal disease.”
Personal details
Gunther Marsche works in the Institute of Experimental and Clinical Pharmacology at the Medical University of Graz where he conducts research on the dysfunctional metabolism of cholesterol and the development of arteriosclerosis. He received the Otto Kraupp Prize in 2011 for his Habilitation (professorial thesis), which is awarded for the best medical Habilitation in Austria.





